Fitbit! Apple! Who is the Smartwatch Winner? Read on!
By Stephen H. Paul, Ph.D., Temple University
The race between Apple and Fitbit for dominance in health technology is on. Who will be the winner? Will there be other contestants?
The Hiatus Is Over
This series of essays will start a cycle of blogs examining the heart-rate and pulse-monitoring capabilities of two leaders in this field. Back issues of these and future articles will be available online. These leaders market Fitbit and Apple smart watches for runners. Product evaluations of all types of running gear will continue to be discussed.
Introduction
Fitbit is constantly updating its operating systems that function on the Android and Apple OS systems. They seem to have almost many updates as The Weather Channel and that is imperative. This is excellent service. Fitbit’s superb technical assistance program is included in the price of their equipment.
Both firms are extremely active in pulse monitoring.
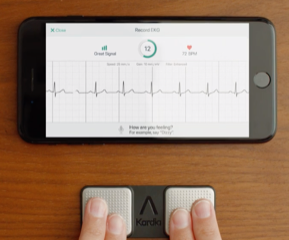
Apple is the first of the technology firms to have a precertification of the Apple Watch series 5 for directly monitoring irregular heartbeats (arrhythmias). Fitbit does not have their own software approved at this time; however, the aftermarket accessory from AliveCor for $99 is called KardiaMobile is shown in this blog. Check with the manufacturer to make sure that your smartphone, computer or tablet will be compatible. Runners who have an earlier series of the Apple Watch may purchase a KardiaBand to replace the strap on earlier Apple Watch Series 1 – 3. These smartwatches all require the watch OS 5.1. The KardiaBand is NOT cleared for ECG usage for the original Apple Watch Series. The band is available at AliveCor for $99. (https://www.alivecor.com) These units will produce a medical acceptable electrocardiogram (EKG or ECG) in 30 seconds. Note: these are two acceptable abbreviations for the term electrocardiogram. The units do not give off electronic radiation.
The FDA is part of the United States Department of Health and Human Services. Its obligation is to protect the public health by assuring the safety, effectiveness, and security of human and veterinary drugs, vaccines, and other biological products for human use, and medical devices. The agency also is responsible for the safety and security of our nation’s food supply, cosmetics, dietary supplements, products that give off electronic radiation, and for regulating tobacco products.
The ECG app approved by the government for Apple is a software-only mobile medical application intended for use with the Apple Watch to create, record, store, transfer, and display a single-channel electrocardiogram (ECG) similar to a Lead I (one) ECG. The ECG app determines the presence of atrial fibrillation (AFib) or sinus rhythm on a classifiable waveform. The ECG app is not recommended for users with other known arrhythmias.
The ECG app is intended for over-the-counter (OTC) use. The ECG data displayed by the app is intended for informational use only. The end user should not interpret or take clinical action based on the device output without consulting a qualified healthcare professional. The ECG waveform is meant to supplement rhythm classification for the purposes of discriminating AFib from normal sinus rhythm. It is not intended to replace traditional methods of diagnosis or treatment.
This app is not intended for use by people under 22 years old.
Both organizations know that early victories with killer applications help to reaffirm the illusion of dominance. It is definitely too early to declare a winner in the health-technology marathon. The first one to cross the finish line is not necessarily the victor. This contest is more like the Olympics with many leaders in numerous events. Additional smartwatch manufacturers and electronic firms are joining the race this year. They will provide a plethora of enhancements and new apps. Initially, the firms should be providing the basics of tracking the pulse of the beating heart up to continuous 24-hour monitoring and the ability graphically to show the results.
The FDA (Food and Drug Administration) of our government is taking an approach toward digital health technology. Their goal after reviewing systems for software design, validation, and maintenance of the technology, is to determine with the developer, if the organization meets quality standards. If they do, then the FDA will pre-cert (precertify) the specific firm(s) as shown in the Proposed Key Components of a Future Pre-Cert Program. Their action would potentially result in less information being submitted to this agency for a new digital-health tool. The pilot participants represent a wide variety of companies and technology in the digital-health sector, including small startups, large companies, high-risk, and low-risk firms. The participants selected in alphabetical order are Apple, Fitbit, Johnson & Johnson, Pear Therapeutics, Phosphorus, Roche, Samsung, and two additional California firms (Tidepool and Verily).
You may submit comments to the public docket at www.regulations.gov using docket number FDA-2018-N-4751 by April 22. The comments for the vision for medical device safety to protect patients and spur innovation of new products that are safer, more effective, and address unmet medical needs.
The federal government agency is considering the new smartwatches as “direct-to-consumer medical devices.” These include the Apple Watch Series 4 and numerous Fitbit models including the Fitbit Ionic, Fitbit Versa, Fitbit Blaze, Fitbit Charge 3, and the Fitbit Inspire HR. The increasing adoption of this new class of consumer-grade wearable devices monitoring heart rate and blood pressure as well as technology combining data from digital scales, sleep information, diet, exercise, and environmental issues should prove valuable for health care practitioners diagnosing and managing the heart health of runners.
The newer direct-to-consumer medical devices with their created apps measuring the pulse rate of the heart and electrocardiographic signals determine if there is the likelihood of one or more heart rhythm problems (heart arrhythmias). An arrhythmia results when the electrical impulses that coordinate heartbeats do not work properly. This could cause the heart to beat too fast, too slow, or too irregular.
Basic Heart Terms
Palpitations
Palpitations are used to describe the feeling of your heart beating. Runners have said that this sensation feels like a fluttering in your chest, or the heart pounding. Some also describe it as feeling like a thud or movement in your chest, which can be experienced sometimes in your neck or through your ear when lying down. Palpitations can be felt whether the heart is beating normally, quickly, slowly, or irregularly. Unfortunately, some of us may not be able to feel these changes. Palpitations are common and for most of us they are harmless. They can be a nuisance and be very unpleasant. They can happen when achieving a PR in a 10K race or really exerting yourself on an uphill challenge. Being very anxious or stressed can also bring them out. Night running could also create these uncharacteristic symptoms in otherwise hearts of normal athletes.
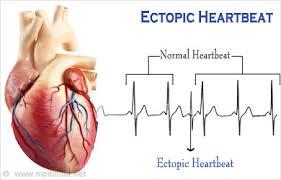
Ectopic Heartbeats
Some arrhythmias may feel like the heart beats are faster than normal while others may feel the beat seems to be fluttering. Ask your health professional to show you how to properly feel your heart beating. One effective way is by pressing one or two fingers on your carotid artery. There is a carotid artery on either side of your neck. Never press on both arteries at the same time as you could faint or feel very dizzy.
Ectopic heartbeats are early (premature) or extra heartbeats. You have the sensation of feeling your own heart beating. Ectopic means out of place. Ectopic beats happen when cells away from the heart’s own natural pacemaker get a little excited (or irritable) and release an electrical signal, causing an “extra” or early heartbeat. There is often a tiny pause after the extra beat, giving you the sensation of a missed beat.
Ectopic beats are premature atrial contractions (PAC) and/or premature ventricular contractions (PVC). Premature atrial contractions (PAC) cause an early electrical impulse in the atria (upper chambers) of the heart. Premature ventricular contractions (PVC) are an early electrical impulse in the ventricles, which are the lower chambers of the heart.
Another type of arrhythmia is known as AFib that many people have seen, heard, and/or read on social media. AFib is atrial fibrillation (a skipped heartbeat and then feeling a thud or thump. It may be followed by the heart racing for a long period of time. Another symptom might be the feel of heart palpitations or fluttering or jumping of the heart. There are numerous other symptoms include sweating, and/or chest pain. This could result in a heart attack, stroke, blood clot or other conditions impacting on the heart or blood vessels (cardiovascular) and possibly the life of the individual.
Ectopic beats are usually harmless and do not mean you have a serious heart condition. They generally require no treatment unless they occur very often or are very severe. If you are not sure, always check the symptoms out with your health professional. The life you save can be your own or a significant other.
The next article will include some medical knowledge on understanding what the ECG shows, how the Kardia units work, and understanding the information direct-to-consumer smartwatches watches can realistically provide.




Leave a comment